Tag: Sirtex
Twelve-month DOORwaY90 results demonstrate complete local tumour control for HCC
Twelve-month results from the DOORwaY90 study, the first pivotal, prospective, multicentre US trial of Y-90 selective internal radiation therapy (SIRT) using partition dosimetry, show...
Sirtex receives expanded CE mark approval for SIR-Spheres Y90 resin microspheres
Sirtex Medical today announced that it has received an expanded CE mark approval for SIR-Spheres yttrium 90 (Y90) resin microspheres for the treatment of...
LAVA trial demonstrates high clinical success for EVOH
Results from the prospective, multicentre Liquid Embolization of Arterial Haemorrhages in Peripheral Vasculature (LAVA) study evaluating the Lava (Sirtex) ethylene vinyl alcohol (EVOH) liquid...
Efficacy and flexibility of SIRT with SIRSpheres® for treatment of HCC...
This advertorial is sponsored by Sirtex Medical.
Highlights:
This four-page supplement shares insights given by key opinion leaders in selective internal radiation therapy (SIRT) using SIR-Spheres...
EmboCept® S degradable starch microspheres show high efficacy and good tolerability...
This advertorial is sponsored by Sirtex.
Interventional radiologists Thomas Albrecht (Vivantes Clinic Neukölln, Berlin, Germany) and Roberto Iezzi (Policlinico Universitario Fondazione Agostino Gemelli, Rome, Italy)...
Sirtex Medical announces commercial availability of liquid embolic system
Sirtex Medical, a leading manufacturer of interventional treatment solutions, has announced the commercial availability of the LAVA liquid embolic system, the first and only...
Sirtex Medical announces Magle Group EmboCept S collaboration agreement
Sirtex Medical, a leading manufacturer of interventional treatment solutions, has announced the establishment of a collaboration and exclusive worldwide distribution agreement with Magle Group,...
How SIR-Spheres® Y-90 resin microspheres can improve patient outcomes, offer greater...
This advertorial is sponsored by Sirtex Medical.
Two experts in the field of interventional radiology, Bernhard Gebauer (head of interventional radiology at Charité – Universitätsmedizin...
Sirtex Medical and China Grand Pharmaceutical announce successful first administration of...
Sirtex Medical (Sirtex) has announced that the first procedure of selective internal radiation therapy (SIRT) using SIRSpheres Y-90 resin microspheres in China was successfully...
DOORwaY-90 study for SIR-Spheres therapy as first-line treatment for hepatocellular carcinoma...
The first patient has been enrolled in DOORwaY-90, a study evaluating the safety and efficacy of selective internal radiation therapy (SIRT) using SIR-Spheres Y-90...
First patient enrolled in pivotal LAVA study
The first patient has been enrolled in the LAVA study to evaluate the safety and effectiveness of the BlackSwan Lava liquid embolic system (LES)...
DOORwaY90 study launches: The first prospective, multicentre US-based trial for registration...
The US Food and Drug Administration (FDA) have approved the DOORwaY90 study, a trial evaluating the safety and efficacy of selective internal radiation therapy...
Sirtex Medical announces strategic investment in BlackSwan Vascular
Sirtex Medical (Sirtex) has announced a strategic investment in BlackSwan Vascular (BlackSwan), a private company developing therapies in endovascular embolization. The investment provides resources...
SIR-Spheres Y-90 resin microspheres meet drug registration requirements in China
Sirtex Medical and its shareholders, China Grand Pharmaceutical and Healthcare Holdings Limited (CGP) have been issued a “Notice of Drug Clinical Trial Approval” by...
The largest European cohort on the treatment of liver cancer with...
The largest European-wide observational study on the treatment of primary and metastatic liver tumours with SIR-Spheres microspheres (Sirtex) will present its data on safety...
Sirtex Medical completes enrollment of landmark US RESiN registry
Sirtex Medical reports that enrollment in the US RESiN registry has reached a milestone of 1,650 patients and 12.8 months of follow-up, allowing for...
Sirtex Medical partners with Magle Chemoswed to develop imaging agent
Sirtex Medical has announced a collaboration with Magle Chemoswed, a Sweden-based contract development and manufacturing organisation.
The development and commercial agreement grants Sirtex global use...
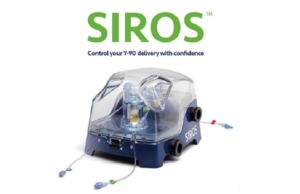
Sirtex Medical launches SIROS system for SIR-Spheres resin microspheres delivery
A new system designed to deliver Yttrium-90 (Y-90) resin microspheres during targeted liver cancer therapies, SIROS (Sirtex) has launched commercially.
A company press release states:...
Sirtex Medical announces collaboration with MIM Software
Sirtex Medical has announced a collaboration with MIM Software. Under the terms of the agreement, Sirtex’s sales teams in the USA, Europe, and other...
SIRT approved for routine commissioning on the NHS
National Health Service (NHS) England have announced that selective internal radiation therapy (SIRT) using yttrium-90 (Y-90) microspheres will now be routinely commissioned for NHS...
Lessons from randomised controlled trials utilising SIRT in liver cancer
A recent session at CIRSE 2018 sought to go beyond the headline findings from a number of recent randomised controlled trials (RCTs) in interventional...
Sirtex prefers US$1.4 billion CDH-CGP bid to Varian’s acquisition offer
Sirtex has announced entering into a binding scheme implementation deed with CDH Genetech (CDH) and China Grand Pharmaceuticals and Healthcare Holdings Limited (CGP). It...
Establishing the evidence base for SIRT in Interventional Oncology
Multidisciplinary oncology thought leaders discuss the personalisation of SIRT, new and recent evidence for the minimally invasive procedure over sorafenib in advanced HCC, the...
SIRT fails to extend survival in the SORAMIC study palliative cohort
The SORAMIC study, presented at The International Liver Congress, shows that the addition of selective internal radiation therapy (SIRT) to sorafenib in patients with...
Varian signs agreement to acquire Sirtex
Varian Medical has announced it has signed an agreement to acquire all the outstanding shares of Sirtex Medical for A$28 per share in cash....
REsect study shows SIRT makes metastatic colorectal cancer patients more likely...
Metastatic colorectal cancer patients treated first-line with SIR-spheres Y-90 resin microspheres more likely to become candidates for potentially curative liver surgery, the REsect study...
SIRT significantly better tolerated than sorafenib, but does not increase overall...
The 459-patient randomised controlled SARAH study shows that local treatments of advanced or inoperable hepatocellular carcinoma with selective internal radiation therapy (SIRT) did not...
New NCCN guidelines recommend SIRT for the treatment of metastatic colorectal...
Sirtex has announced that SIR-Spheres Y-90 resin microspheres have been included as a Category 2A recommended treatment in the latest National Comprehensive Cancer Network...
New ESMO biliary cancer guidelines indicate radioembolization as an option for...
New European Society of Medical Oncology (ESMO) biliary cancer guidelines indicate yttrium-90 (Y-90) therapy as an option for post-chemotherapy treatment of intrahepatic cholangiocarcinoma.
Sirtex has announced...