Reflow Medical has announced that 12-month results from the DEEPER REVEAL clinical trial were presented for the first time at the Society of Interventional Radiology (SIR) annual scientific meeting (11–15 April, Toronto, Canada).
The data were presented by Constantino Peña (Miami Cardiac and Vascular Institute at Baptist Hospital, Miami, Florida) on behalf of the study’s principal investigators, Mahmood K Razavi (St Joseph Heart and Vascular Center, Orange, USA); S Jay Mathews (Manatee Memorial Hospital, Bradenton, USA); Jihad A Mustapha (Advanced Cardiac & Vascular Centers for Amputation Prevention, Grand Rapids, USA), and the DEEPER REVEAL trial investigators.
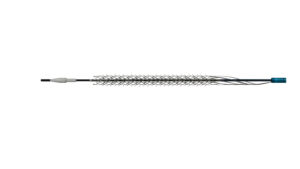
DEEPER REVEAL is a prospective, non-randomised, multicentre study evaluating use of the Spur peripheral retrievable stent system following predilatation in patients with chronic limb-threatening ischemia (CLTI) and below-the-knee (BTK) arterial disease. A total of 130 patients are enrolled across 49 US centres. In May 2025, the Spur stent system was granted US Food and Drug Administration (FDA) De Novo clearance in a high-risk, long-lesion population.
At 12 months, vessel-related outcomes in appropriately selected vessels (core lab-measured reference vessel diameter ≥2.5mm, as defined per study protocol and indication for use) and patient-centred outcomes were assessed.
“These 12-month results demonstrate sustained clinical outcomes in a challenging population of patients with severe CLTI and complex BTK disease,” said Peña. “At 12 months, we observed primary patency of 78% and freedom from clinically driven target lesion revascularisation of 83.1% in appropriately sized target vessels.”
Patient-centred outcomes improved from baseline, including complete paired wound healing in 84.3% of patients, symptomatic improvement measured by Rutherford class in 80% of patients, and significant gains in vascular quality of life.
Peña continued: “Importantly, these findings compare favourably to well-established historical plain balloon angioplasty data and support the role of retrievable scaffold therapy with the Spur stent system as a valuable treatment approach in this high-risk population.”
Reflow describes the Spur stent system as a “unique” clinical solution for the treatment of de novo or restenotic lesions following predilatation in patients with infrapopliteal arterial disease. It provides temporary mechanical scaffolding to support the vessel wall and facilitate blood flow. The Spur stent incorporates radially expandable spikes for controlled lesion penetration and modification, improving vessel compliance and reducing vessel recoil. Following treatment, the device is fully retrieved, preserving future treatment options.
“The DEEPER REVEAL 12-month results reinforce the clinical value of Reflow Medical’s retrievable scaffold therapy in addressing the significant unmet need in patients with CLTI,” said Carolyn Mascho, director of clinical affairs at Reflow Medical.
Isa Rizk, CEO and co-founder of Reflow Medical, added: “Building on the Breakthrough Device designation and FDA De Novo clearance for Spur, these data support our efforts to expand access to innovative treatment options for physicians treating complex BTK disease.”