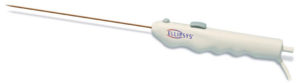
Two devices for creating minimally invasive dialysis access—the Ellipsys vascular access system (Avenu Medical) and the WavelinQ 4F system (BD)—demonstrated high rates of technical success and low rates of complications, according to a new study published in the Journal of Vascular and Interventional Radiology. In addition, both technologies enabled patients to start dialysis sooner compared to surgery, offering significant quality-of-life benefits.
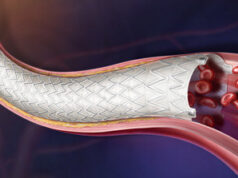
For patients with end-stage renal disease (ESRD) who require haemodialysis, the preferred type of vascular access is an arteriovenous fistula (AVF)—a permanent connection between a vein and artery in the arm. Until recently, the only way to create an AVF was through a surgical procedure that subjects patients to discomfort, longer recovery times and delayed dialysis.
Both the Ellipsys and WavelinQ devices offer a nonsurgical alternative by using a minimally invasive approach to create a percutaneous arteriovenous fistula (pAVF). The new single-centre study, led by vascular surgeon Robert Shahverdyan (Vascular Access Center, Hamburg, Germany), compared the outcomes for 100 patients who underwent pAVF procedures (65 Ellipsys and 35 WavelinQ) from December 2017 to December 2019.
The Ellipsys system had a technical success rate of 100%, and only 27.7% of patients required secondary interventions following fistula creation. The WavelinQ system demonstrated similar outcomes, with a technical success rate of 97% and 26.5% of patients requiring secondary interventions. Overall, 79.5% of Ellipsys patients and 58% of WavelinQ patients were able to successfully begin dialysis with their fistula. At 12 months, 82% of Ellipsys fistulas were still functional, compared to 60% of WavelinQ fistulas.
“For every patient, my goal is to create the best possible dialysis access with the fewest possible interventions,” said Shahverdyan. “Our data show that with careful planning, both systems can safely and quickly create percutaneous fistulas with a high rate of success, though the Ellipsys system appears to create more durable access that is also easier to repair if that becomes necessary.”
Compared to surgical fistulas, both devices reduced the time to dialysis (cannulation). The mean time to cannulation for WavelinQ was 90 days, compared to 60 days with Ellipsys. In fact, 75% of Ellipsys fistulas were ready for cannulation at just four weeks, with a small number of patients ready for early cannulation within a few days of fistula creation, Shahverdyan notes.
“This study confirms other newly published data showing how much faster we can start using the Ellipsys percutaneous fistula for dialysis—a finding that has significant patient safety benefits,” said interventional radiologist Jeffrey Hull (Richmond Vascular Center, Richmond, USA). “The sooner a patient can start dialysis with their fistula, the sooner we can get them off such riskier dialysis access methods as central venous catheters, which are associated with much higher rates of infection and other complications.”
Hull is the lead author on a recent study of Ellipsys pAVF that reported an average time to dialysis of 66 days. Vascular surgeon Alexandros Mallios (Hôpital Paris Saint-Joseph, Paris, France), recently published data showing that patients were able to begin using their pAVF for dialysis an average of 4 weeks after creation, with six percent of fistulas becoming functional in as little as 2 weeks.
The studied technologies differ significantly in their design, pAVF creation technique and fistula location. The single-catheter Ellipsys system uses ultrasound guidance and thermal energy to create the fused and permanent connection between the perforating vein and the proximal radial artery; no radiation or contrast media are required. The WavelinQ device is a two-catheter system that uses radio-frequency energy and fluoroscopy to create an aligned slit fistula. Differences between the systems can affect patient eligibility for the procedures based on anatomic constraints.
“Based on our preoperative ultrasound evaluations, we found that 65% of patients were eligible for an Ellipsys pAVF, compared to just 27% with the WavelinQ system,” said Shahverdyan. “By knowing that more than twice as many patients will be able to undergo the procedure, Ellipsys makes it more feasible for a center to offer this endovascular approach to creating dialysis access.”
Cleared by the US Food and Drug Administration (FDA) in 2018 for patients with end-stage renal disease, Ellipsys is the first significant innovation in AVF creation in over 50 years. It transforms a complex surgery into a minimally invasive procedure that can be performed in a hospital outpatient setting, ambulatory surgery center or physician’s office. Since 2015, nearly 3,000 patients worldwide have undergone the Ellipsys procedure. Recently published long-term data show that 92% of Ellipsys fistulas are still functional after two years. The study also found high levels of patient satisfaction with the procedure.