The US Food and Drug Administration (FDA) have approved AngioDynamic’s investigational device exemption (IDE) application for NanoKnife Irreversible Electroporation “Direct IRE Cancer Treatment” clinical study (DIRECT).
The DIRECT Study supports a proposed expanded indication for the NanoKnife system in the treatment of stage III pancreatic cancer. In January 2018, the FDA granted the company’s NanoKnife system a breakthrough device designation under the 21st Century Cures Act.
According to a press release, the NanoKnife system is a next-generation ablative technology that physicians have identified as an innovative treatment for pancreatic cancer. AngioDynamics state that this is evidenced by the 42 publications that document more than 800 patients who have been treated for stage III pancreatic cancer with NanoKnife technology from 2012 to 2019.
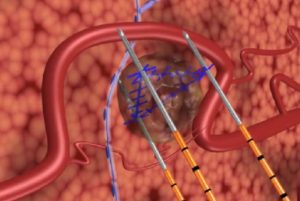
The NanoKnife system received 510(k) clearance from the FDA for the surgical ablation of soft tissue in 2008. Unlike other ablative technologies, the NanoKnife system utilises low-energy, direct-current electrical pulses to permanently open pores in target cell membranes and does not rely on thermal effects, the press release explains. These permanent defects in the cell membranes result in cell death. The treated tissue is then removed by the body’s natural processes in a matter of weeks, mimicking natural cell death.
AngioDynamics state that their DIRECT study will feature a “comprehensive data collection strategy” that will provide meaningful clinical information to healthcare professionals, support a regulatory indication for the treatment of stage III pancreatic cancer, and facilitate reimbursement for hospitals and treating physicians. The study is classified as a Category B IDE by the FDA, allowing participating sites to obtain coverage for procedures performed as well as related routine costs.
The DIRECT study comprises a randomised controlled trial at up to 15 sites and a real-world evidence, next-generation registry (RWE) at up to 30 sites, each with a NanoKnife system treatment arm and a control arm. AngioDynamics expects each NanoKnife arm to consist of approximately 250 patients with an equal number of control patients. The primary endpoint of the study is overall survival.
The randomised controlled trial component of the DIRECT study will illustrate the promise of the NanoKnife system by isolating variables in a controlled setting, says the press release. Recognising that results from a hyper-controlled setting do not always translate to clinical practice, the study also includes a RWE component to provide clinicians, patients, and payers with data generated in a real-world setting.
As part of the DIRECT clinical study, AngioDynamics is launching AngioDIRECT.com to facilitate the enrollment of participants. The online platform will provide patients and their families with information about pancreatic cancer and details about the study. It will also feature a physician locator to help prospective participants and referring healthcare professionals identify clinical study locations.
“Existing evidence that has accumulated over the last 10 years has shown that irreversible electroporation is an effective adjunctive treatment for patients with locally advanced pancreatic cancer,” comments Robert C G Martin, co-principal investigator of the DIRECT study and surgical oncologist at the University of Louisville, Louisville, USA. “We anticipate that this trial will demonstrate how the NanoKnife system can be utilised to enhance the quality of life for patients with stage III pancreatic cancer.”
“I have had promising experience utilising irreversible electroporation as a treatment option for pancreatic cancer patients,” says Govindarajan Narayanan, co-principal investigator of the DIRECT study and chief of Interventional Oncology at the Miami Cancer Institute. “We expect that the results of this trial will lead to a widely available alternative treatment option for advanced pancreatic cancer patients.”
There are approximately 57,000 new cases and 46,000 estimated deaths from pancreatic cancer in the USA annually. Total deaths due to pancreas cancer are projected to increase dramatically to become the second leading cause of cancer-related deaths before 2030. The mortality rate is high due to the aggressive nature of the disease and lack of early warning signs, and less than 20% of patients are candidates for surgical resection at time of diagnosis. Approximately 35–40% of patients will present with stage III and 45–55% with metastatic disease. Regardless of the stage of pancreatic cancer, it is one of the least survivable cancers, and survival rates have not improved substantially for more than 40 years. For all stages combined, the five-year relative survival rate is 8% and, for those with advanced disease at the time of diagnosis, the five-year survival rate remains at 3%.
There are limited treatment options for stage III and IV disease, with chemotherapy and/or radiotherapy considered the standard of care. There have been advancements in both techniques, but this has come at the cost of greater toxicity, limiting the number of patients that are candidates for treatment.










Interested in the nano-nif method
Before surgery using a robotic method
Gleason rank 4 + 3 = 7 What are the success rates and if you can talk to people who have passed this method.