Tag: Ivascular
LUMIFOLLOW registry shows sustained three-year performance of Luminor in femoropopliteal disease
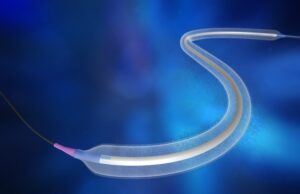
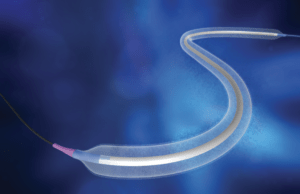
iVascular has revealed the three-year follow-up results from the LUMIFOLLOW registry, a large real-world study evaluating the safety and efficacy of the Luminor drug-coated...
BARISTA trial demonstrates positive one-year results in treatment of illac artery...
iVascular has announced the one-year results from the BARISTA trial, the first clinical trial with Restorer bare metal stent evaluating its safety and efficacy...
First-in-human study demonstrates complete vascular occlusion with peripheral liquid embolic agent
“Our conclusion is: Amber is safe, efficacious and might change the way we technically embolize some of our indications today,” were the closing remarks...
Luminor drug-coated balloon launched in Japan
iVascular has announced that its Luminor drug-coated balloon (DCB) is now available in Japan.
“This approval from the MHLW is evidence of the quality,...
iVascular initiates Opal study with new embolic liquid
iVascular has announced the initiation of Opal, the first-in-human study with the new embolic liquid Amber, based on a radiopaque biocompatible copolymer. Opal is...
First results of SOL Japan highlight “extraordinary performance” of Luminor DCB...
iVascular has announced that "outstanding" results from the SOL Japan study were recently presented at the Japan Endovascular Treatment (JET) conference (14–16 June, Fukuoka,...
iCover’s radiopaque markers and full encapsulation with small profiles help it...
Vicente Riambau (Barcelona, Spain) describes the unique features and improvements of the iCover balloon expandable covered stent, notably its low profile, radiopaque markers, and...
LUMIFOLLOW one-year outcomes show efficacy for femoropopliteal lesions
Luminor (iVascular) drug-coated balloon (DCB) has confirmed its excellent safety and efficacy in femoropopliteal lesions with new study LUMIFOLLOW, a recent press release states.
iVascular...
Full cohort results confirms efficacy of Luminor drug-coated balloon for BTK...
iVascular reflect on the Luminor drug-coated balloon (DCB) in below-the-knee (BTK) outcomes obtained in the BIBLIOS trial one-year follow-up presented at the Paris Vascular...
Five-year RCT data provide upbeat message on Luminor paclitaxel-coated balloon’s safety...
NOTE: This video is ONLY available to watch in selected countries and geographies
The EffPac trial was a prospective, multicentre, randomised controlled trial (RCT)...
New TINTIN data indicate longer term success of DCB and stent...
NOTE: This video is ONLY available to watch in selected countries and geographies
The combined use of a drug-eluting device and a vascular scaffold...
iVascular receives CE mark for iCover covered stent
iVascular has received CE mark approval for its balloon-expandable covered stent, iCover, a press release from the company states.
Balloon-expandable covered stents are commonly used...
Episode 1: Luminor DCB in the femoropopliteal segment: Outstanding safety and...
NOTE: This video is ONLY available to watch in selected countries and geographies
Koen Deloose (Dendermonde, Belgium) moderates a three-part Vascular News series about the Luminor...
Episode 2: Experience of Luminor DCB in the below-the-knee segment
NOTE: This video is ONLY available to watch in selected countries and geographies
Koen Deloose (Dendermonde, Belgium) moderates a three-part Vascular News series about the Luminor...
Episode 3: Not all DCBs are the same: Luminor TransferTech nanotechnology...
NOTE: This video is ONLY available to watch in selected countries and geographies
Koen Deloose (Dendermonde, Belgium) moderates a three-part Vascular News series about the Luminor...
“No signal of increased risk of long-term mortality” with paclitaxel-coated Luminor...
An ad hoc, two-year analysis of the EffPac study found no signal of increased risk of long-term mortality, nor any adverse events, within two...
MERLION trial six-month outcomes presented at VIVA late-breaking trial session
Six-month clinical outcomes of the MERLION trial were presented as part of the VIVA Late-Breaking Clinical Trials Livesteam (25 June), by Tjun Tang (Singapore...
iVascular launches Sergeant peripheral support catheter
iVascular recently announced the launch of its Sergeant peripheral support catheter. Sergeant is a CE marked over-the-wire catheter, indicated for patients with peripheral arterial...
Luminor DCB now reimbursed in France
Luminor 18 and Luminor 35 drug-coated balloons (DCB; iVascular) are now listed on the French LPPR list for de novo lesions of the femoropopliteal...
TINTIN trial shows “impressive” results at six months
The six-month results of TINTIN (Treatment with the lumINor DCB and The IVolutioN self-expanding stent) trial have been revealed at the annual meeting of the Cardiovascular and...
Experts discuss latest results from EFFPAC and TINTIN trials presented at...
NOTE: ONLY intended for healthcare professionals outside of the USA.
Ulf Teichgräber (Jena, Germany) and Koen Deloose (Dendermonde, Belgium) discuss the 24-month results of the...
BIBLIOS trial has obtained ethics approval and initiated patient enrolment
The BIBLIOS trial has received Ethics Committee (EC) approval, and enrolled its first patient. This announcement comes from Wouter Lansink, who will be treating this...
Enrolment complete in T.I.N.T.I.N. study
iVascular has announced the completion of enrolment of patients in its T.I.N.T.I.N. (Treatment with the Luminor DCB and the iVolution self-expanding stent) study.
The effectiveness...
CX 2018: Ulf Teichgräber discusses EffPAC trial 12-month results
Ulf Teichgräber, Jena, Germany, discusses the EffPAC trial 12-month results which were presented for the first time at CX 2018
Luminor DCB shows high clinical effectiveness in six-month EffPAC data
The six-month results from the full clinical cohort of the EffPAC randomised study were presented in the drug-coated balloon (DCB) abstracts session at the...
Ivolution now available in 200mm length devices
Ivascular has announced the product range extension of its self-expandable stent, iVolution, to 200mm length, in various diameters.
Ivolution is a nitinol endovascular stent, combining...