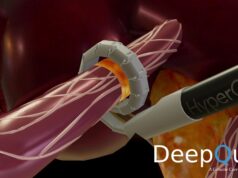
SoniVie today announced that on 30 May this year the first patient was treated with its renal artery denervation Tivus therapeutic intravascular ultrasound technology, as part of the recently US Food and Drug Administration (FDA) investigational device exemption (IDE)-approved REDUCED1 pilot study to treat hypertension.
A company press release details that the procedure was successfully performed with Tivus at the Kaplan Medical Center (Rehovot, Israel) by Michael Jonas, principal investigator of the REDUCED1 study.
Jonas commented: “We were able to use the Tivus catheter to rapidly and efficiently perform ultrasound-based renal denervation in our patient suffering from uncontrolled hypertention. The Tivus catheter’s excellent ease of use and its ability to denervate at significant depth of the perivascular space allowed for a short, simple and straightforward procedure. We will closely follow our patient and look forward to continuing enrolment in this exciting trial.”
Renal denervation with Tivus is a minimally invasive procedure that uses high-frequency non-focused ultrasound energy to ablate nerves in the renal artery, SoniVie advises in the press release. This causes a reduction in the nerve activity, which may decrease blood pressure. The procedure is designed for patients who suffer from resistant hypertension.
“Millions of people worldwide suffer from resistant hypertension which substantially increases the risk of heart attack, stroke and kidney failure. We are genuinely happy for this important step forward. There is a lack of effective therapeutic solutions for these patients” says Christian Spaulding, CMO, SoniVie.
“This procedure represents a great fulfilment for SoniVie team, whose expertise and achievements have been focusing on developing Tivus technology for the renal denervation indication, both providing a promising treatment to the hypertensive patient and a high performing, easy to use device to the physician”, says Tomaso Zambelli, CEO, SoniVie.