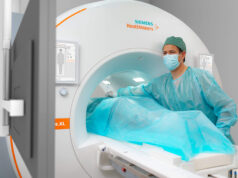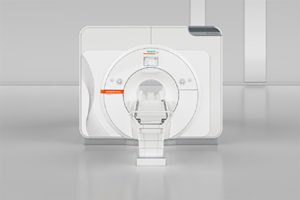
Siemens Healthineers has received the CE mark for the 7 Tesla magnetic resonance (MR) scanner Magnetom Terra, making it the first-ever ultra-high-field MR scanner to be approved for clinical use, a press release from the company says.
Clinicians in Europe can now use the system for neurological and musculoskeletal examinations in routine clinical applications.
“Thanks to our long-standing commitment and experience with ultra-high-field MRI as well as our extensive innovation network of collaboration partners around the world, we are now establishing 7T imaging in the clinical routine with Magnetom Terra”, says Christoph Zindel, senior vice president and general manager of Magnetic Resonance Imaging at Siemens Healthineers. “Having now gained approval for clinical use, we are expanding the scope of diagnostic MRI – 15 years after 3T scanners first became established. With this new clinical field strength, we can achieve a new level of detail in anatomy and function, helping further pave the way for precision medicine,” says Zindel. “I am convinced that Magnetom Terra will help grow the footprint of 7 Tesla in research and clinical application, allowing us to further explore new territories in MRI.”
“This ability to use 7 Tesla technology in the clinical realm is a breakthrough into broader clinical 7 Tesla MR applications. This CE certificate is now well-supported by the remarkable progress with respect to novel radiofrequency technologies and MR methods that will make whole-body MRI at 7 Tesla feasible in the future. It will definitely foster the translation of research into clinical applications,” says Siegfried Trattnig, director of the MR Centre of Excellence at the Medical University of Vienna, Austria, “And, the higher signal to noise ratio, the improved contrast, and non-proton MR at 7T will translate into better spatiotemporal resolution, but even more importantly into enhanced biochemical and metabolic tissue characterisation en route to clinical noninvasive personalised medicine.”