
A new study is underway at Emory Healthcare, Atlanta, USA, to evaluate the use of cryoablation to treat mild to moderate obesity.
The clinical trial, called “Percutaneous image-guided cryoablation of the vagus nerve for management of mild-moderate obesity” will assess the safety of cryoablation of the posterior vagus nerve.
This study seeks to assess the efficacy of cryoablation for the treatment of mild to moderate obesity and test the safety of this procedure. The vagus nerve transmits hunger signals from the stomach to the brain, and in response transmits stomach expansion signals from the brain to the stomach. Investigators believe that by interrupting this communication, participants could experience less hunger and in turn, lose weight.
The two-year study will enrol 20 patients, and participants will serve as their own control group. Patients with a body mass index of 30 to 35 who are unable to lose weight with diet changes or exercise are eligible for inclusion if they have no coagulopathy.
The primary outcome measures include record of the number of adverse events which occur within the first 24 hours following cryoablation; clinical signs or symptoms of infection, haemorrhage that occur outside of the time periods specified for cryoablation procedure events; and significant adverse events include life-threatening adverse events and death upto two years.
J David Prologo, director of Interventional Radiology Services at Emory Johns Creek Hospital, and assistant professor in the Department of Radiology and Imaging Sciences in Emory’s School of Medicine, is the principal investigator of this interventional study.
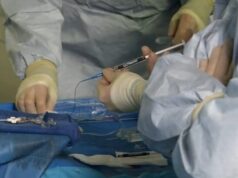
“We use image guidance to insert a cryoablation probe near the stomach, targeting the nerve that sends hunger signals to the brain,” says Prologo.
The trial will examine patients using self-reported questionnaires regarding physical activity, appetite and living habits as well as data measurement.
Participants will attend five visits for evaluation and doctors will follow their progress for six months after the procedure. The outpatient cryoablation procedure takes about an hour.
Endocare is funding this clinical trial.
For information regarding enrolment, please contact Maria Rivas: [email protected]










where can I find help to lose weight and keep it off I was looking into radiology intervention procedure if possible