 Guerbet has been granted approval for an indication extension in the UK, where Lipiodol Ultra Fluid is the only oil-based contrast medium indicated for hysterosalpingography (HSG) in women undergoing infertility workup. New clinical evidence showing that an HSG with Lipiodol Ultra Fluid enhances pregnancy rates has been recognised by the UK’s Medicines and Healthcare products Regulatory Authority (MHRA). MHRA’s endorsement of evidence-based clinical practice confirms the benefit of Lipiodol Ultra Fluid in fertility management, a Guerbet press release claims.
Guerbet has been granted approval for an indication extension in the UK, where Lipiodol Ultra Fluid is the only oil-based contrast medium indicated for hysterosalpingography (HSG) in women undergoing infertility workup. New clinical evidence showing that an HSG with Lipiodol Ultra Fluid enhances pregnancy rates has been recognised by the UK’s Medicines and Healthcare products Regulatory Authority (MHRA). MHRA’s endorsement of evidence-based clinical practice confirms the benefit of Lipiodol Ultra Fluid in fertility management, a Guerbet press release claims.
One in seven couples face fertility issues in the UK. The fertility workup, during which HSG is performed, is a key step in patient care management.
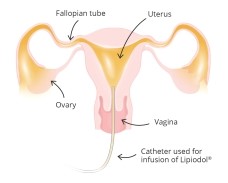
The technique involves the slow infusion of Lipiodol Ultra Fluid into the uterus, using X-ray monitoring to assess the uterine cavity and tubes.
More importantly, women who underwent an HSG with Lipiodol Ultra Fluid had enhanced pregnancy outcomes:
- In a single centre randomised trial: in a group of women with unexplained infertility or endometriosis who received Lipiodol Ultra Fluid (n=73), twice as many became pregnant within six months compared to women who did not benefit from the procedure (n=85). The pregnancy rate was 38.4% and 16.5% respectively (p=0.002).
- In a multicentre randomised trial: in a group of women who underwent an HSG with Lipiodol Ultra Fluid (n=554), 37% more became pregnant within six months compared to women who received an HSG with a water-based contrast medium (n=554). The pregnancy rate was 39.7% and 29.1%, respectively (rate ratio (RR): 1.37 [1.16; 1.61]; p<0.001).
- In a meta-analysis of randomised controlled trials: in a group of women who underwent an HSG with Lipiodol Ultra Fluid, 42% more became pregnant within six months compared to women who received a water-based contrast medium. The overall odds ratio (OR) for pregnancy was significantly in favour of HSG with Lipiodol Ultra Fluid: odds ratio of 1.42 [1.10; 1.85] when compared to HSG with a water-based contrast medium (six studies, n=2,598).
“This is great news for UK patients experiencing difficulties conceiving, as it brings them additional hope by enhancing their chances of falling pregnant at an early stage of their fertility journey. As with any procedure, patients should be aware of any associated risks,” says Gwenda Burns, chief executive of Fertility Network UK.
“Our clinical experience of Lipiodol Ultra Fluid has showed us that its use is similar to water-based contrast media and does not alter the patient experience during the exam. However, it is noteworthy that long term follow-up studies showed that performing an HSG with Lipiodol Ultra Fluid reduces the median time to pregnancy by around four months, as compared to an HSG with the reference contrast media,” comments Colin Duncan (Royal Infirmary Edinburgh, Edinburgh, UK; chair of the Society for Reproduction and Fertility).
“At Imperial College Healthcare NHS Trust, we have performed more than 10,000 HSGs over the last few decades. The evidence-based results from the H2Oil study encouraged our HSG and fertility teams to use Lipiodol Ultra Fluid for HSG in selected patients. After several months of use, we can confirm that an HSG with Lipiodol Ultra Fluid is an exciting and valuable technique in the management of patients being investigated for fertility issues in our centre. Our teams at Imperial College Healthcare NHS Trust will soon to be involved as investigators in on-going randomised controlled clinical trials evaluating the performance of Lipiodol Ultra Fluid for HSG in extended populations undergoing fertility workup,” adds Anne Hemingway (Imperial College Healthcare NHS Trust, London, UK).
The UK is the sixth European country to grant approval for HSG in women undergoing infertility workup after Ireland, Hungary, The Netherlands, the Czech Republic, and Denmark. Lipiodol Ultra Fluid is also currently approved for HSG in ten additional countries (the USA, including Puerto Rico, Canada, Argentina, Turkey, South Africa, Japan, Taiwan, Thailand, Australia and New Zealand).
In 2016, the Guerbet Group launched its Women’s Health franchise with the ambition to improve the quality of infertility management and breast cancer surgery.










