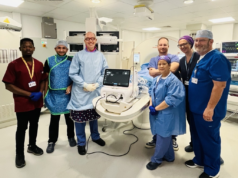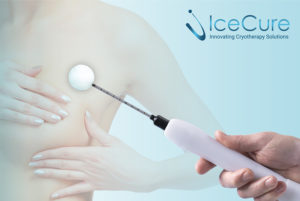
IceCure Medical has announced in a press release that Health Canada, the Canadian government’s regulatory agency, has approved IceCure’s ProSense system, disposable cryoprobes, and introducers as cryosurgical tools for indications including:
- Tumours—ablation of benign and malignant tumours of the lung, liver, kidneys, and musculoskeletal system, and benign tumours of the breast;
- General surgery;
- Palliative intervention; and
- Other surgeries.
“As we continue to gain traction in the USA, this approval in Canada will create synergies for us in the broader North American market. We have been directly engaging with healthcare providers through education, hands-on training seminars, marketing, and sales in the USA and we will expand these activities into Canada. We have already been contacted by multiple Canadian healthcare providers with requests to purchase and implement ProSense,” stated IceCure Chief Executive Officer Eyal Shamir.
Canada’s healthcare system is ranked as one of the top in the world, with total health spending estimated at $331 billion in 2022.
IceCure’s cryoablation system currently has regulatory approval for various indications in 15 countries, including in the USA, Europe, and China.