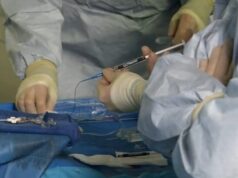
Fluidx Medical Technology, has recently announced the oversubscribed closing of the first tranche of the Series A financing round. The Series A was led by a large multinational strategic investor and joined by the existing investor base as well as several new investors. The funding will be used to support prospective clinical trials, including the pivotal trial, as well as preparation for market entry.
“GPX has been quite simple to prepare, deliver and control,” said Andrew Holden (director of Interventional Radiology, Auckland City Hospital, Auckland, New Zealand). “The material is very radiopaque during delivery and has demonstrated excellent distal penetration, particularly important in our tumor embolization cases.”
Embolic devices are widely used to stop blood flow for targeted treatment of hypervascular tumors, vessel malformations, internal bleeds, and other cancer-related uses. Challenges with embolic devices today can include complicated preparation processes, issues with visibility during device delivery, reliance on the body’s coagulation situation, and lack of complete penetration of vessel beds and tumor feeding vessels.
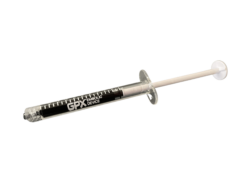
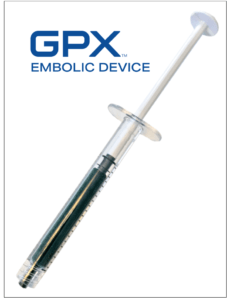
The GPX Embolic Device is an innovative embolic designed for simple preparation and quick material delivery. GPX technology is a low viscosity, aqueous-based solution in a syringe that solidifies into a durable embolus upon delivery without polymerization or dimethyl-sulfoxide (DMSO) precipitation. GPX is designed to occlude blood vessels independent of a patient’s coagulation situation. Unlike competitive technologies that require special preparation materials and time, the GPX device is packaged ready-to-use in a syringe, requires less than 30 seconds of tableside preparation by the clinician, and may be delivered through standard catheters or small microcatheters (no complex mixing systems or special delivery catheters are necessary).