 New long-term data presented at Charing Cross (CX) demonstrated the safety of BIOTRONIK’s Passeo-18 Lux paclitaxel drug-coated balloon (DCB) for the treatment of peripheral artery disease (PAD) in infrainguinal arteries.
New long-term data presented at Charing Cross (CX) demonstrated the safety of BIOTRONIK’s Passeo-18 Lux paclitaxel drug-coated balloon (DCB) for the treatment of peripheral artery disease (PAD) in infrainguinal arteries.
The independent analysis was led and presented by Ron Waksman, Clinical Professor of Medicine, Georgetown University and Associate Director of Cardiology, MedStar Washington Hospital Center, Washington D.C., USA. Individual patient-level data were pooled from three trials in the Passeo-18 Lux DCB clinical programme: the BIOLUX P-I and P-II randomised controlled trials (RCTs) and the BIOLUX P-III all-comers registry. Five-year follow-up data from 943 patients treated with Passeo-18 Lux DCB and 66 patients treated with percutaneous transluminal angioplasty (PTA) were evaluated.
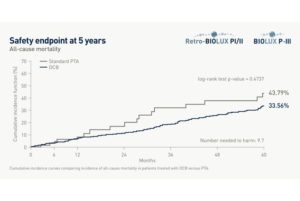
The analysis showed that the use of the Passeo-18 Lux paclitaxel DCB in infrainguinal arteries was not associated with an increase in all-cause mortality at five years compared to standard PTA (33.56% for DCB versus 43.79% for PTA; P=0.47).
“Our recent analysis adds to the body of long-term evidence supporting paclitaxel-coated balloons, which are an essential and safe tool for the treatment of patients with PAD who are presenting with increasingly challenging disease and are often at high risk of restenosis,” commented Ron Waksman on the results. “In line with previous clinical data, the Passeo-18 Lux DCB has been shown to be safe at five years when compared to PTA.”
These results confirm the previous analysis of this dataset, published last year, which reported similar mortality at one year irrespective of the treatment with DCB or PTA and clear reduction in the incidence of the composite endpoint of any amputation and target lesion/vessel revascularisation compared to standard PTA. Other recent data have also shown similar mortality rates in patients treated with paclitaxel-coated balloons compared to PTA.
“We are pleased to contribute evidence that helps to address concerns about the safety of paclitaxel-coated technologies and confirm Passeo-18 Lux’s excellent safety record. We are continuing to invest in DCB clinical research including the upcoming BIOPACT RCT, which will further support evidence-based decision making,” said Alexander Uhl, president of vascular intervention at BIOTRONIK.
The BIOPACT RCT clinical trial, which completed enrolment last year, will provide a head-to-head comparison of Passeo-18 Lux and IN.PACT Admiral DCBs. The primary endpoint will be presented upon completion of 12-month follow-up.











