This case report has been sponsored by BD.
Panagiotis M Kitrou, consultant interventional radiologist, and Dimitrios Karnabatidis, professor of Interventional Radiology, both at Patras University Hospital, Patras, Greece, report on a case in which they successfully used a COVERATM Vascular Covered Stent (Bard, now BD) in combination with a high pressure balloon to treat two significant stenoses.
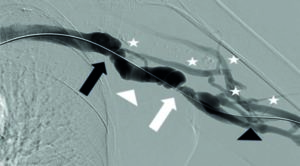
A 62-year-old patient with a brachio-axillary arterio-venous graft (AVG) was referred from his dialysis center with signs of inadequate dialysis. The patient’s venogram revealed a significant stenosis at the graft-vein anastomosis and a second stenosis just distal with presence of extensive venous-tributary network (Figure 1).
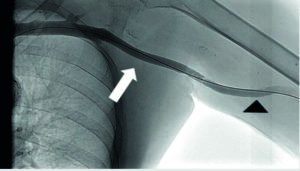
A 7mm diameter, 80mm length DORADO® High Pressure Balloon (BDPI, Tempe, Arizona, USA) was used to “beat” the stenosis. A flared 8mm diameter 80mm length COVERA™ Vascular Covered Stent was placed to cover both lesions. The final result is shown in Figure 2.
More than 50% of stenosis in AV Access Grafts present within 1cm around the venous anastomosis.1 Therefore, most new technologies have been tested and evaluated in this specific AV Graft segment. A study of paramount importance, published by Haskal et al in 2010, demonstrated a significant patency improvement over plain balloon angioplasty in both Access Circuit and Target Lesion when the FLAIR® Endovascular Stent Graft was used.2 Since then, several studies supported the above mentioned results showing consistency of data.3, 4 Until today, stent grafts are the only devices to provide significant atency improvement at six months compared to plain balloon angioplasty in multicentre randomized controlled trials.
The COVERA™ Vascular Covered Stent is used in our department for the treatment of venous-graft anastomotic stenosis. With our experience of more than 100 devices, the COVERA™ Covered Stent has proven to be an excellent and durable solution for the treatment of those resistant and frequently re-occurring lesions. Additionally, the ease of the deploying mechanism is an advantage because it can be controlled by fellows that have limited interventional experience. Data from the AVeVA study (prospective, multicentre, non-randomised, single-arm clinical study) showed a freedom from primary safety events rate of 96.4% and a six-month target lesion primary patency rate of 70.3% for the COVERA™ Covered Stent.
Our algorithmic approach suggests using a Covered Stent at the venous-anastomotic stenosis as a primary treatment in grafts older than 18 months and for patients with symptoms recurring within a short period of time (<3 months) after balloon angioplasty.
References
1. Kanterman RY, Vesely TM, Pilgram TK, Guy BW, Windus DW, Picus D. Dialysis access grafts: anatomic location of venous stenosis and results of angioplasty. Radiology. 1995;195(1):135-9.
2. Haskal ZJ, Trerotola S, Dolmatch B, Schuman E, Altman S, Mietling S, et al. Stent graft versus balloon angioplasty for failing dialysis-access grafts. The New England journal of medicine. 2010;362(6):494-503.
3. Haskal ZJ, Saad TF, Hoggard JG, Cooper RI, Lipkowitz GS, Gerges A, et al. Prospective, Randomized, Concurrently-Controlled Study of a Stent Graft versus Balloon Angioplasty for Treatment of Arteriovenous Access Graft Stenosis: 2-Year Results of the RENOVA Study. Journal of vascular and interventional radiology: JVIR. 2016;27(8):1105-14 e3.
4. Karnabatidis D, Kitrou P, Spiliopoulos S, Katsanos K, Diamantopoulos A, Christeas N, et al. Stent-grafts versus angioplasty and/or bare metal stents for failing arteriovenous grafts: a cross-over longitudinal study. Journal of nephrology. 2013;26(2):389-95.
The opinions and clinical experiences presented herein are for informational purposes only. The results from this case study may not be predictive for all patients. Individual results may vary depending on a variety of patient specific attributes. The physician has been compensated by BD for the time and effort in preparing the above case study for BD’s further use and distribution.










