This case report is sponsored by BD.
Ralf Langhoff, chief, Angiology/Vascular Medicine, Sankt Gertrauden-Krankenhaus, Berlin, Germany, reports on a case in which his team used a novel 0.035” UltraScore™ Focused Force PTA Balloon (Bard, now BD) as an adjunctive therapy with the LUTONIX® drug coated balloon (DCB) to recanalise a chronic total occlusion (CTO) in the superficial femoral artery (SFA).
A 68-year-old female was referred to our vascular center for intermittent claudication primarily in the right leg (Rutherford category 3). She is a current smoker with additional risk factors for atherosclerosis, which are arterial hypertension and dyslipidemia with high cholesterol levels. She had a pain free walking distance below 50 meters, with an Ankle Brachial Index (ABI) of 0.63 on the right side.
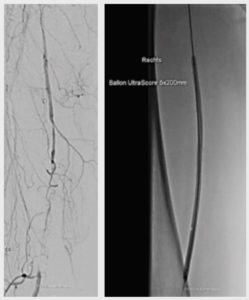
After the referring angiologist performed a duplex examination, the test revealed a diseased SFA from the origin of the artery with an occlusion at the distal end. The total length of the diseased segment was 260mm (Fig. 1).
To avoid long segment stenting and to prepare the vessel for better drug-uptake, we intended to intraluminally cross the occlusion and follow with a dedicated scoring balloon for percutaneous transluminal angioplasty (PTA) and vessel preparation.
We used a cross-over access from the left transfemoral with a 6F Fortress Introducer Sheath, 45cm length (Biotronik) and passed the lesion with a 0.035” GLIDEWIRE®, 260cm length (Terumo). As support, we used a 4F multi-purpose catheter, TEMPO AQUA® (Cordis, now Cardinal Health).
Subsequently, vessel preparation was performed with the novel UltraScore™ 5mmx200mm scoring balloon (Bard/BD), which runs on a 0.035” wire (Fig. 2). The intention of vessel preparation is to minimize the risk of dissections, and maximize luminal gain to prepare the vessel for local drug delivery and/or stents.
The balloon was inflated slowly, which allowed the wires to provide concentrated force on the diseased vessel wall. This focused force dilatation led to a controlled plaque fracture, improved luminal gain. Additionally, the objective was to limit risk of major dissections (SOGA 2018) as severe dissections are related to negative long-term outcomes in femoro-popliteal interventions (FUJIHARA 2017). After seeing a very good and promising result after the use of the UltraScore™ balloon, we decided to use DCBs to deliver paclitaxel to the entire lesion.
Furthermore, there is the idea that by using scoring elements during vessel preparation the drug-uptake of DCBs can be increased and optimized (CREMERS 2013). We used two 5mmx150mm LUTONIX® DCBs (Bard/BD) with a 1cm overlap to avoid a geographical miss. We chose a longer balloon inflation time of three minutes because we revealed a short, minor dissection in the former occluded part of the lesion. We decided against provisional stenting because results of the large DCB trials reported strong effects of positive remodeling after application of paclitaxel to the vessel wall via a balloon (TEPE 2013).
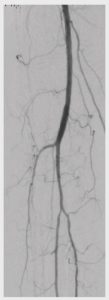
Control angiography demonstrated a very good result, with a small minor dissection which was not flow limiting in several projections.
The intervention was finished without any complications and a good outflow (Fig. 3). We used a MYNXGRIP® device (Cordis/Cardinal Health) for hemostasis and vessel closure.
After the case, the patient was free of any ischemic pain while walking.
References
1. Soga Y, Ando K. Effect of an NSE PTA balloon in experimental lesion models. Cardiovasc Interv Ther. 2018 Jan;33(1):35–39.
2. Fujihara M, et al. Angiographic dissection patterns and patency outcomes after balloon angioplasty for superficial femoral artery disease. J Endovasc Ther. 2017;24:367–75.
3. Cremers B, et al. Inhibition of neo-intimal hyperplasiain porcine coronary arteries utilizing a novel paclitaxel-coated scoring balloon catheter. Catheter Cardiovasc Interv. 2014 Dec 1;84(7):1089–98.
4. Tepe G, et al. High-grade, non-flow limiting dissections do not negatively
impact long-term outcome after paclitaxel-coated balloon angioplasty:
an additional analysis from the THUNDER study. J Endovasc Ther. 2013;20:792–800.