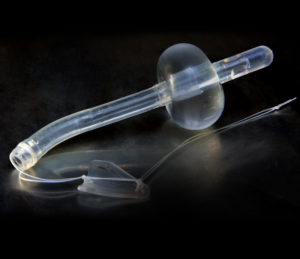
SRS Medical Systems has announced that it has received CE mark approval for use of its Spanner temporary prostate stent in the European Economic Area (EEA). Spanner is an alternative to both indwelling (Foley) and intermittent urinary catheters for male patients with bladder outlet obstruction.
“The Spanner is proven to have significant impact on medical outcomes, and often has a transformational impact on patient quality of life,” said Lee Brody, CEO of SRS Medical. “We are excited to receive this approval, and we look forward to working with our European partners to deliver The Spanner to the patients that will benefit from it the most.”
Spanner is a completely internal device that alleviates bladder outlet obstruction while maintaining continence. It allows patients to naturally fill and empty their bladders, often without device awareness. The device has many clinical applications including:
- Relieving lower urinary tract symptoms in patients in temporary urinary retention, including those recovering from surgical procedures;
- Reducing infection risk and medical complication in patients awaiting procedures for benign prostatic hyperplasia;
- Offering long-term symptom relief for patients unfit for prostate procedures; and predicting benign prostatic hyperplasia procedure outcomes in high-risk surgical candidates.
In the USA, Spanner is approved by the FDA for a single 30-day period in a limited patient population. In January 2016, SRS Medical received FDA investigational device exemption (IDE) approval for a confirmatory safety study to expand the labelling of Spanner.
Spanner is an FDA-approved class III medical device intended to maintain urine flow and allow for voluntary urination in certain male patients experiencing lower urinary tract symptoms. The device consists of two anchors and a silicone tube that reduces resistance in the bladder neck and prostatic urethra without stenting the external sphincter. Spanner is placed blindly without anaesthesia, in a procedure similar to the placement of a Foley catheter. It is reimbursed under CPT Code 53855, and is the only prostate stent on the US market, a press release from SRS Medical says.













