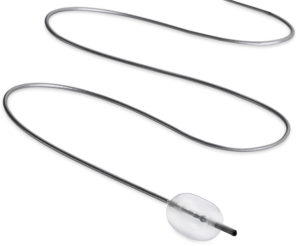
Embolx, a new medical device company out of Silicon Valley, has been given the European CE mark for its latest Sniper balloon occlusion microcatheters. The devices are used to help deliver embolic agents in various procedures, including the treatment of tumours, uterine fibroids, and enlarged prostate.
The Sniper balloon occlusion microcatheters were recently cleared in the USA, gaining Food and Drug Administration (FDA) approval earlier this year.
The device occludes the vessel to alter blood flow-dynamics using pressure-directed embolization. It is currently used for the treatment of cancerous tumours in the liver and other organs, enlarged prostate (benign prostatic hyperplasia), and uterine fibroids.
“Our next generation devices incorporate experience from more than 1,000 Sniper balloon occlusion microcatheters used to date,” says Michael Allen, president and CEO of Embolx. “Now, interventional radiologists can do everything they would do with standard microcatheters, but with the added benefits of balloon occlusion. This is a big step forward and provides physicians with the most advanced transarterial delivery system for tumour and prostate treatment.”
The Sniper comes in three lengths—110cm, 130cm, and 150cm—enabling access to femoral and radial arterial sites. Improvements in the microcatheter’s atraumatic tip allow for smooth entry and easy visualisation thanks to the radiopaque material, a press release informs. The Sniper is also compatible with conventional diagnostic catheters, guidewires, and embolic agents, so physicians do not require additional accessories.
This European clearance follows the news in April this year that the US National Institute of Health (NIH)’s National Cancer Institute awarded a US$2 million small business innovation research grant to Embolx to fund further research and development of the company’s Sniper balloon occlusion microcatheter.











