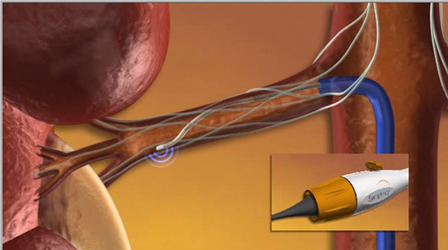
Final clinical outcomes from the first and longest-running renal denervation clinical study with the Symplicity renal denervation system (Medtronic) were presented during the European Society of Cardiology congress and have been accepted for publication in The Lancet.
Eighty eight treatment-resistant hypertension patients were available for 36-month evaluations following treatment with the Symplicity renal denervation system. These 88 patients demonstrated sustained reductions in blood pressure year-over-year with an average reduction of -32/-14mmHg [p<0.01]. Of these 88 patients, approximately 50% achieved goal of systolic blood pressure.
“We are pleased to find that we are seeing sustained and significant blood pressure reductions overall in all patients who reached the three year time point following their denervation procedure. Achieving a goal of below 140mmHg in about half of these patients is impressive considering that these patients had very high baseline blood pressures despite being on multiple pharmaceutical agents,” said Henry Krum, principal investigator of SYMPLICITY HTN-1 and chair of Medical Therapeutics, professor of Medicine and director of the Monash Centre of Cardiovascular Research and Education in Therapeutics, Melbourne, Australia, who presented the data at ESC. “These were patients who were out of hypertension treatment options before they received renal denervation, so reductions of blood pressure of this magnitude may dramatically decrease their risk for stroke, heart attack, heart failure and kidney disease in the years to come.”
SYMPLICITY HTN-1 is a series of pilot studies at 19 centres in Australia, Europe and the United States. The open-label studies enrolled 153 patients with treatment-resistant hypertension, defined as having a systolic blood pressure ≥160mmHg while taking 3 antihypertensive drugs at optimal dosages, including a diuretic. The patients consented to be followed for either one, two, or three years after treatment with renal denervation. Follow-up is now complete for the 88 patients who were followed to three years. SYMPLICITY HTN-1 is the largest cohort of patients with the longest follow-up data for renal denervation to date.
Patients treated with renal denervation experienced consistent reductions in blood pressure regardless of advanced age, the presence of diabetes or impaired baseline renal function. Safety follow-up between 24 and 36 months demonstrated continued stable renal function; two orthostatic hypotension events in one subject resolved with medication changes, and one renal artery stenosis at 24 months, was possibly related to the renal denervation procedure. Adverse events due to co-morbid diseases such as infection and non-renal surgical complications were also reported.










