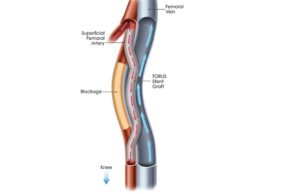
Tag: PQ Bypass
Endologix announces acquisition of PQ Bypass
Endologix recently announced it has completed the acquisition of PQ Bypass, a medical technology company pioneering a first-of-its-kind technology that addresses an unmet need...
DETOUR I two-year outcomes: “Excellent” functional improvement in complex PAD cohort
During a late-breaking data session at this year’s Vascular Interventional Advances annual meeting (VIVA 2020; 6–8 November, virtual), Ehrin Armstrong (University of Colorado, Denver,...
PQ Bypass completes enrolment in DETOUR II pivotal study
PQ Bypass has announced enrolment of the final subject in the company’s DETOUR II investigational device exemption (IDE) clinical trial. This milestone occurs only...
PQ Bypass receives FDA breakthrough device designation for the Detour system
PQ Bypass has announced that they received breakthrough device designation from the US Food and Drug Administration (FDA) for the Detour system. The Detour...
DETOUR I 12-month data show promising durability of bypass system
Results from the DETOUR I trial evaluating the DETOUR System (PQ Bypass) for percutaneous bypass showed promising 12-month durability for patients with extremely long...
PQ Bypass receives IDE approval to initiate pivotal DETOUR II clinical...
PQ Bypass has received conditional approval of its investigational device exemption (IDE) from the US Food and Drug Administration (FDA) to initiate the pivotal...