Tag: lysis
New mechanical thrombectomy systems unlock possibilities for the treatment of venous...
This advertorial, sponsored by Inari Medical, is only available in selected countries and geographies.
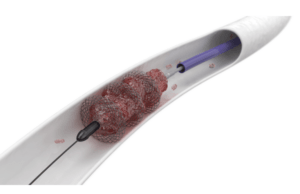
Mechanical thrombectomy with the ClotTriever and FlowTriever systems (Inari Medical) opens...
BTG receives US FDA 510(k) clearance for Ekos control unit 4.0
BTG has announced that US FDA 510(k) clearance has been granted to the Ekos control unit 4.0.
The Ekos system includes an ultrasonic device...