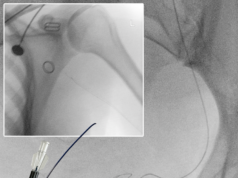
 The results from a small prospective, randomised single-centre trial, the PREMIERE trial, show that in patients with unresectable hepatocellular carcinoma, there is a significantly longer time to progression with yttrium-90 (Y-90) radioembolization than with conventional transarterial chemoembolization (cTACE).
The results from a small prospective, randomised single-centre trial, the PREMIERE trial, show that in patients with unresectable hepatocellular carcinoma, there is a significantly longer time to progression with yttrium-90 (Y-90) radioembolization than with conventional transarterial chemoembolization (cTACE).
The study further finds that improved time to progression with Y-90 therapy did not result in a significant difference in overall survival, when comparing the two treatments, but the researchers posit that the results translate to Y-90 treatment achieving better local tumour control and lower dropout from transplant listing. CTACE is the standard of care for patients ineligible for radiofrequency ablation as included in the study.
“The PREMIERE trial prospectively compared chemoembolization and radioembolization in a randomised fashion and this is what physicians have wanted to see for a long time. We started the study in 2009 and had set the sample size at 124 patients. However, what has happened over the last eight years is that radioembolization as a treatment modality has now been adopted routinely and as a result, patients no longer want to be randomised. Out of the 124 patents, we only enrolled 45. We still analysed the data from the 45 patients in a blinded manner. Despite the relatively small sample size, the results were extremely positive. The hazard ratio was 0.13, in other words the risk of progression in the radioembolization arm was 87% less than in the cTACE arm, which is very compelling. Very rarely in cancer studies do you have such low hazard ratios. Despite the small sample size, these statistics are very significant. This is a post hoc analysis but the clinical findings of segmental Y-90 results are very impressive” Riad Salem, chief, Vascular and Interventional Radiology and director, Division of Interventional Oncology, Department of Radiology, Northwestern University, Chicago, Illinois told Interventional News.
The PREMIERE study set out to determine the comparative effectiveness of cTACE vs. Y-90 radioembolization for patients with unresectable hepatocellular carcinoma considered ineligible for ablation in a randomised setting.
“We included patients who were not radiofrequency ablation candidates. If a patient is not a radiofrequency ablation candidate, the staged migration concept advocates that they move up to the next stage and are treated with cTACE. The progression is usually: radiofrequency ablation, cTACE, sorafenib and then, supportive care. cTACE is also the standard of care if the disease is intermediate. Hence, we took everyone eligible for cTACE and then randomised them to either receive cTACE or Y-90. As a result, this cohort consists of mostly BCLC A and some BCLC B patients, with solitary and multifocal disease and these patients showed a dramatic result,” Salem explained.
The investigators carried out a single-blind, randomised phase II single-centre study from October 2009 to September 2015. They enrolled 45 patients (median age 63.8 years; 72% male; 53% solitary hepatocellular carcinoma) with Barcelona Clinic Liver Cancer (BCLC) stage A or B, ECOG 0-2, Child-Pugh A or B, and no systemic therapy. The patients were randomly assigned to either receive cTACE or Y-90 therapy using glass microspheres.
Patients were followed for clinical and laboratory adverse events using version four of the Common Terminology Criteria for Adverse Events (CTCAE) as well as baseline and follow-up imaging at one month, and every three months thereafter. The primary outcome was time-to-progression evaluated on intention-to-treat from the date of randomisation. Secondary outcomes were safety, imaging response rates (WHO and EASL criteria), and overall survival.
Results from the study showed that serious (Grade 3+) clinical toxicities and objective response rates on follow-up imaging were similar across the two therapies. “In total, 12 patients progressed, 15 died prior to liver transplant, and 19 received liver transplants. Median time to progression was just under six and half months in the cTACE group, and not reached in the Y90 group (p=0.002). Median overall survival censored to liver transplant was 17.7 months in the cTACE group and 18.6 months in the Y-90 group, respectively (p=0.94). In BCLC A patients, 6/15 cTACE vs. 13/18 Y-90 received liver transplant (p=0.085) with a trend towards longer median time to transplant at 17.3 months for cTACE vs. 9.1 month after Y-90 (p=0.0579).
Commenting on whether it was possible that Northwestern had a super-optimised Y-90 technique and perhaps a less refined TACE technique, Salem clarified: “We have published on our chemoembolization technique. Our results with 172 patients have been published in Radiology and is one of the largest US cohorts treated with cTACE. This showed that our results are comparable to others. Recently, we published in JVIR our updated data with another 165 patients showing consistent results. So clearly, this shows that our cTACE technique is optimal. Is it possible that we have a hyper improved Y-90 technique? While we have always advocated meticulous technique, if you look at papers from Bruno Sangro’s group in Spain, and other groups in Germany and Vincenzo Mazzaferro’s group in Italy, they all have the same results as us, suggesting the techniques and outcomes have been replicated. Therefore, although those are theories trying to explain away the findings, they are not valid. They can be rebutted easily—our Y-90 results have been replicated and our TACE outcomes are equivalent to others. What we are left with is huge difference between Y-90 and TACE that most people observe clinically.”
How do these results apply in practice? “Now that we have these randomised data, our multidisciplinary group believes they should be acted upon, and that is why we now go straight to Y-90 in this patient population. That is the whole reason to generate data—you should not be paralysed by the findings. So for us, when trying to achieve local tumour control, Y-90 has now been shown to outperform cTACE by a prospective randomised trial,” Salem concluded.