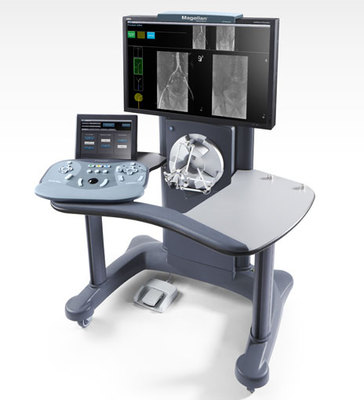
Hansen Medical has announced that it has received 510(k) clearance from the US FDA for its Magellan Robotic System, including the catheter and accessories. The company will commence commercialisation at selected centres in the US immediately, with a full launch expected later in the year.
The Magellan Robotic System is intended to facilitate navigation to anatomical targets in the peripheral vasculature and subsequently provide a conduit for manual placement of therapeutic devices. The system has the potential to provide vascular surgeons and other interventionalists the ability to perform fast and predictable procedures, while allowing the physician to be seated comfortably away from the radiation source, which may reduce radiation exposure and physician fatigue.
“Not only does the Magellan system have the potential to be a significant growth driver for Hansen, it also represents a fundamental step forward in the transformation of vascular intervention using intravascular robotics,” said Bruce Barclay, president and CEO, Hansen Medical.
Alan Lumsden, chair of Hansen Medical’s US Scientific Advisory Board, and chair of the Department of Cardiovascular Surgery and Medical Director of Methodist DeBakey Heart and Vascular Center at The Methodist Hospital, Houston, USA, said: “The Magellan Robotic System is a significant technological advancement that may offer important clinical benefits for physicians performing peripheral interventions. The system provides physicians with independent robotic control of both catheter tips to navigate efficiently through a variety of anatomies and lesions. Our in vitro and in vivo animal studies indicate that using this platform has the potential to increase efficiencies in the interventional lab by shortening procedure times and allowing more predictable interventions. Ultimately, we believe this system may facilitate alternative patient treatment options by enabling robotic endovascular interventions.”
Jean Bismuth, a leading vascular surgeon at the DeBakey Heart and Vascular Center at The Methodist Hospital, Houston, said: “Since the Magellan Robotic System was designed specifically for vascular interventions, it offers excellent catheter stability and precision during the delivery and placement of a variety of therapeutic devices in different anatomic conditions, including various peripheral vascular diseases with tortuous anatomy. Additionally, the Magellan Robotic System may offer physicians less radiation exposure and reduced procedural fatigue due to the remote workstation that allows the physician to be seated comfortably outside the imaging suite.”
The Magellan system received a CE mark in the European Union last year, and the company has already commenced a commercial launch in that region. In addition, the system has been approved in Australia, and is pending approval in Canada.