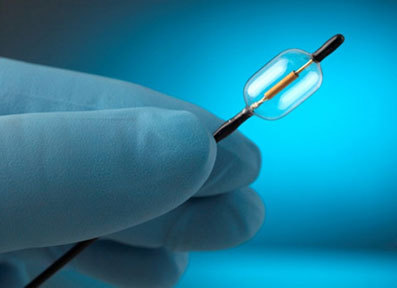
ReCor Medical has announced that it has completed enrolment in the REDUCE 15-patient pilot clinical study of its PARADISE (Percutaneous renal denervation system) ultrasound platform for treatment of resistant hypertension.
Thomas A Mabin,Vergelegen Medi-Clinic, South Africa, principal investigator, will present the current results of the study on 17 May at EuroPCR.
Preliminary clinical data for Paradise were previously reported by Mabin at the recent “TRenD 2012” transcatheter renal denervation scientific meeting. The PARADISE data showed that systolic blood pressure was reduced by an average of 31 mm Hg in 7 patients at 60-days follow-up. The scientific literature demonstrates that only a 5 mm Hg reduction in BP results in a 14% decrease in stroke, a 9% decrease in heart disease, and a 7% decrease in mortality.
“ReCor’s Paradise system is designed to provide a minimally invasive ultrasound therapy to resistant hypertension patients to help reduce their blood pressure,” said Mano Iyer, president and CEO, ReCor Medical. “In the REDUCE study, we have been able to show that our novel Paradise system is capable of safely and quickly denervating patients, resulting in sustained blood pressure reductions out to 6 months. We look forward to Dr Mabin’s reporting of the current results of our REDUCE pilot clinical trial at PCR as we move forward with our launch of our Paradisetm system in Europe.”










