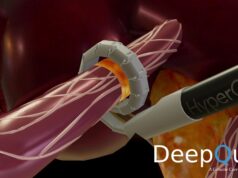
Latest guidelines from the European Society of Cardiology (ESC) on the management of blood pressure and hypertension include a recommendation for the use of renal denervation as a tool to reduce blood pressure.
The treatment—in which energy is targeted through a catheter to the renal nerves to modulate the sympathetic signalling between the kidneys and brain to reduce blood pressure—has been included with a class IIb level of recommendation for treating resistant hypertension in patients with uncontrolled blood pressure, despite the use of three or more blood pressure-lowering drugs.
Additionally, clinicians can consider the interventional treatment in patients with increased cardiovascular risk who have uncontrolled hypertension on fewer than three drugs.
In both instances the guidelines stipulate that the treatment is contingent on patients expressing a preference for renal denervation after a shared risk-benefit discussion and multidisciplinary assessment, and that the procedure is performed at a medium-to-high volume centre. Furthermore, the therapy is not recommended for patients with highly impaired renal function (eGFR <40mL/min/1.73m2) or secondary causes of hypertension.
During a question and answer session following the presentation of the new guidelines at the 2024 ESC congress (30 August–2 September, London, UK), co-chair of the writing committee that drafted the document, John William McEvoy (University of Galway School of Medicine, Galway, Ireland), explained why there is greater emphasis on additional pharmacological interventions—such as the diuretic medication spironolactone or beta blockers—ahead of the device-based treatment in the algorithm for treating resistant hypertension.
“We followed a policy that to meet a class I recommendation or a higher level, we would like to see evidence from trials not just showing a blood pressure-lowering effect but also an impact on outcomes,” McEvoy said. “When it comes to spironolactone and beta blockers, we felt there was sufficient evidence—it wasn’t always direct evidence in the context of resistant hypertension, but in other areas where these agents have shown benefit on cardiovascular disease outcomes.”
McEvoy said that evidence of the efficacy of renal denervation in improving cardiovascular outcomes, on top of its efficacy in reducing blood pressure, would dictate any future changes.
Demonstrating that reductions in blood pressure resulting from the use of renal denervation do lead to improvements in cardiovascular events would likely elevate the therapy within guidelines, Felix Mahfoud (Saarland University Hospital, Homburg, Germany) who has researched the technique extensively tells Cardiovascular News, but the practicalities of conducting such a trial make it unlikely.
“Both randomised controlled trials and real-world registries have demonstrated the blood pressure-lowering efficacy of renal denervation. It is highly likely that these reductions in blood pressure will translate into improved outcomes, including lower rates of stroke, myocardial infarction, heart failure, and other cardiovascular events,” he comments.
“However, from a purist research perspective, proving this association in a randomised controlled trial would be ideal, as it could eventually lead to a class I recommendation in guidelines. Conducting such an outcomes trial in renal denervation, however, would be challenging due to the need for a large sample size (>10,000 patients), long-term follow-up (five years), and the associated high costs.”
Elsewhere, the 2024 guidelines maintain an existing definition for ‘Hypertension’ as a blood pressure measuring ≥140/90mmHg, but introduce a new category of ‘Elevated blood pressure’ which is defined as 120–139/70–89mmHg. This new category is intended to facilitate consideration of more intensive blood pressure treatment targets among persons at increased risk for cardiovascular disease.
Another change sees the introduction of a new systolic blood pressure treatment target range of 120–129mmHg for most patients receiving blood pressure-lowering medication, with the proviso that the new target requires that treatment is well tolerated.
In part to accommodate this new more intensive systolic blood pressure treatment target range, the 2024 ESC guidelines provide stronger recommendations than prior guidelines for the use of out-of-office blood pressure measurements—including ambulatory blood pressure monitors and validated home blood pressure monitors.
The addition of renal denervation—having previously been confined to use solely in the setting of clinical trials—has been described by advocates of the technique as a big step forward for patients requiring additional options to control hypertension.
“Renal denervation has returned to the guidelines and is now recommended for patients with uncontrolled resistant hypertension, as well as for those who are unable or unwilling to tolerate antihypertensive medications”, comments Mahfoud. “This is reassuring news for both physicians and patients. The guidelines also suggest that renal denervation should be considered as an alternative or additive approach to lowering blood pressure, used in combination with lifestyle modifications and antihypertensive drugs.”
ESC’s latest guidelines come shortly after the American Heart Association (AHA) acknowledged renal denervation as a “promising therapeutic approach” for some patients with uncontrolled hypertension in a scientific statement following a review of evidence into the therapy.
The statement was published in the journal Hypertension and recognises in particular that patients with uncontrolled hypertension, those with resistant hypertension or who have multiple medication intolerances, may be the most likely to benefit from the procedure, albeit with the caveat that further research is needed in areas such as patient selection and long-term efficacy.
“The AHA scientific statement presents an intensive review of trial evidence, followed by clinical considerations and practical recommendations for incorporating renal denervation into hypertension treatment programmes,” said Naomi Fisher (Harvard Medical School and Brigham and Women’s Hospital, Boston, USA), a co-author of the statement and consultant for Recor Medical, one of the two companies that has a renal denervation device approved for use by the US Food and Drug Administration (FDA).
“While more research is needed, most, but not all, of the new generation randomised renal denervation control trials reached their primary endpoint, and the procedure carries a favourable safety profile. The AHA statement serves an important role by illuminating renal denervation as a new treatment option for many patients with uncontrolled hypertension, particularly those with resistant hypertension or who are intolerant to multiple medications.”
In November 2023, the field of renal denervation was buoyed by the approval of two devices by the US Food and Drug Administration (FDA)—the Paradise (Recor Medical) and Symplicity Spyral (Medtronic) systems—following a review of evidence from trials assessing both technologies.
ESC’s guideline recommendations add to recent releases from the Society for Cardiovascular Angiography & Interventions (SCAI)—which heralded the “tremendous potential” of renal denervation for the treatment of hypertension—as well as updated arterial hypertension guidelines from the European Society of Hypertension (ESH), stating that renal denervation can be proposed as an adjunctive therapy in select patients with resistant hypertension.